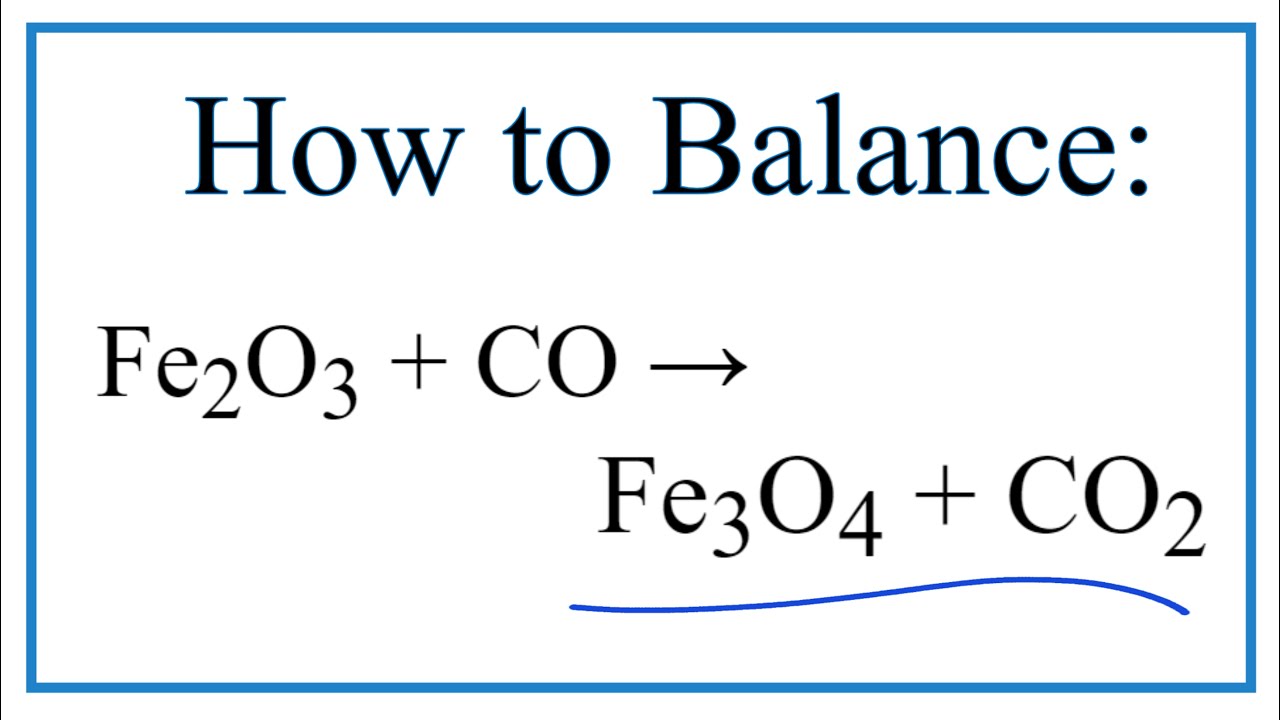
Fe2O3 + 3CO→ 2Fe + 3CO2 FeO + CO→ Fe + CO2 rH = - 26kJ rH = - 16kJ Find H of reaction Fe2O3 + CO→ 2FeO + CO2 rH = ?
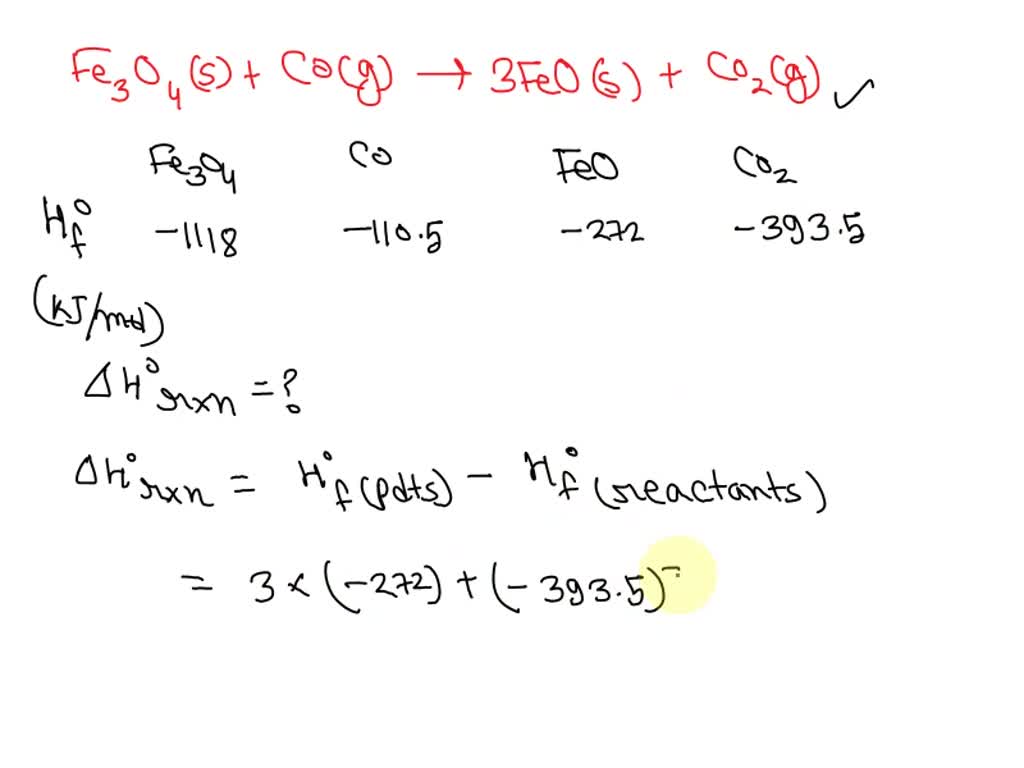
SOLVED: Calculate Horxn for the following reaction at 25.0oC: Fe3O4(s) + CO(g) –> 3FeO(s) + CO2(g) Hfo (kJ/mol) => Fe3O4(s) = -1118, CO(g) = -110.5, FeO(s) = -272, CO2(g) = -393.5

Life Cycle Impact Assessment of Iron Oxide (Fe3O4/γ-Fe2O3) Nanoparticle Synthesis Routes | ACS Sustainable Chemistry & Engineering

Which of the following reactions takes place at higher temperature range ( 900 K - 1500K) in blast furnace?

An Atomic Insight into the Confusion on the Activity of Fe3O4 Nanoparticles as Peroxidase Mimetics and Their Comparison with Horseradish Peroxidase | The Journal of Physical Chemistry Letters

Effect of Rb promoter on Fe3O4 microsphere catalyst for CO2 hydrogenation to light olefins - ScienceDirect

High Conversion to Aromatics via CO2-FT over a CO-Reduced Cu-Fe2O3 Catalyst Integrated with HZSM-5 | ACS Catalysis

SOLVED: Question 29 (2 points) Which of these choices shows the following chemical reaction properly balanced? FezO3 + CO Fe3O4 CO2 FezO3 2CO Fe3O4 2C02 3FezO3 + CO 2Fe3O4 + CO2 None



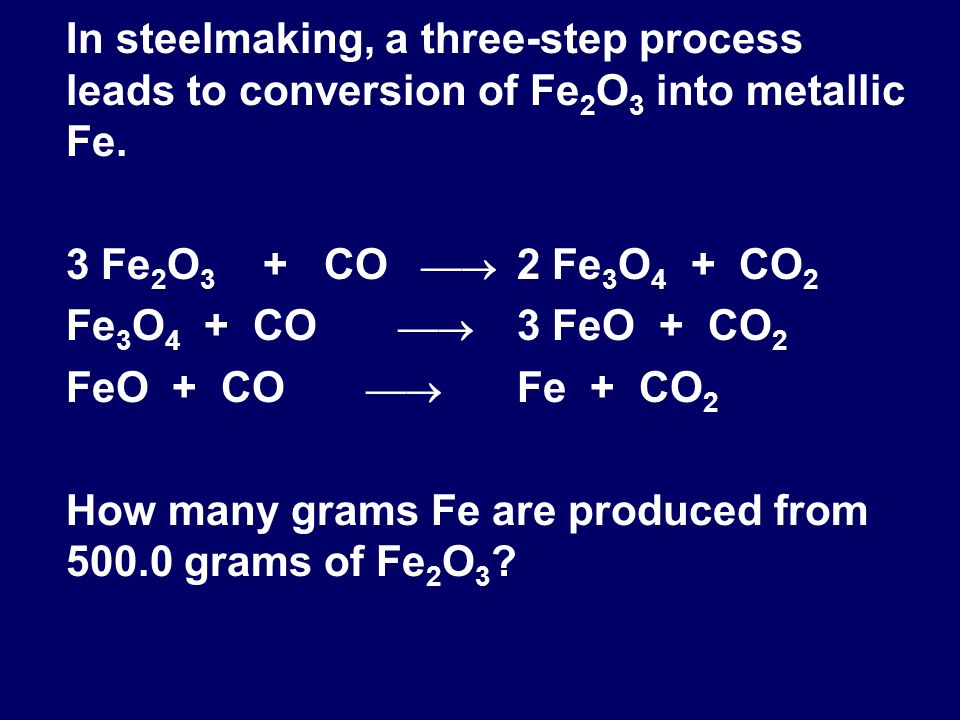



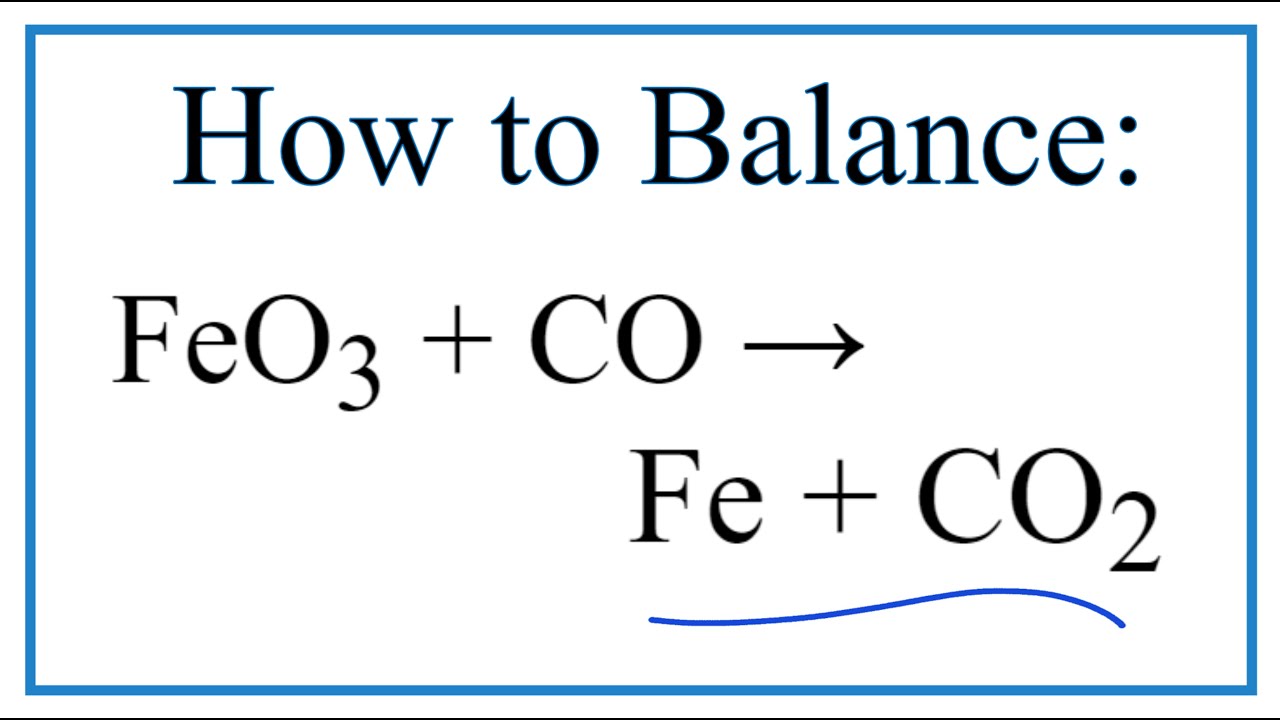


![ANSWERED] Iron oxide ores, commonly a mixture of F... - Inorganic Chemistry ANSWERED] Iron oxide ores, commonly a mixture of F... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/50670046-1658929430.9444451.jpeg)